Fist law of thermodynamics
 All systems transfer energy to their environment through radiation no matter how well insulated they are. The heat from the compressed working fluid is used to warm the building. The entropy of a system at absolute zero is typically zero, and in all cases is determined only by the number of different ground states it has. Work is also equal to the negative external pressure on the system multiplied by the change in volume: Everything outside of the boundary is considered the surroundings, which would include the container itself. If heat flows into a system or the surroundings do work on it, the internal energy increases and the sign of q and w are positive. However, no net energy is created or lost during these transfers.
All systems transfer energy to their environment through radiation no matter how well insulated they are. The heat from the compressed working fluid is used to warm the building. The entropy of a system at absolute zero is typically zero, and in all cases is determined only by the number of different ground states it has. Work is also equal to the negative external pressure on the system multiplied by the change in volume: Everything outside of the boundary is considered the surroundings, which would include the container itself. If heat flows into a system or the surroundings do work on it, the internal energy increases and the sign of q and w are positive. However, no net energy is created or lost during these transfers.

Irene. Age: 29. Hello gentlemen lets spend some time togather I'm discreet, outgoing, fun and will make you feel relaxed and releave some of that every day stress that life can bring with a soft touch, small waist and cute face you'll be sure to come back for more please check out my website for more information






The Three Laws of Thermodynamics
 The Second Law Increased Entropy and the Third Law Zero Entropy at Zero Kelvin are dependent on the First Law and each other. The second law of thermodynamics helps to explain this observation. This released energy appears in the form of heat and light. We will present some simple examples of these laws and properties for a variety of physical systems, although we are most interested in the thermodynamics of propulsion systems and high speed flows. The System and Surroundings One of the basic assumptions of thermodynamics is the idea that we can arbitrarily divide the universe into a system and its surroundings. The Third Law of Thermodynamics The third law of thermodynamics states that the entropy of a system approaches a constant value as the temperature approaches absolute zero.
The Second Law Increased Entropy and the Third Law Zero Entropy at Zero Kelvin are dependent on the First Law and each other. The second law of thermodynamics helps to explain this observation. This released energy appears in the form of heat and light. We will present some simple examples of these laws and properties for a variety of physical systems, although we are most interested in the thermodynamics of propulsion systems and high speed flows. The System and Surroundings One of the basic assumptions of thermodynamics is the idea that we can arbitrarily divide the universe into a system and its surroundings. The Third Law of Thermodynamics The third law of thermodynamics states that the entropy of a system approaches a constant value as the temperature approaches absolute zero.

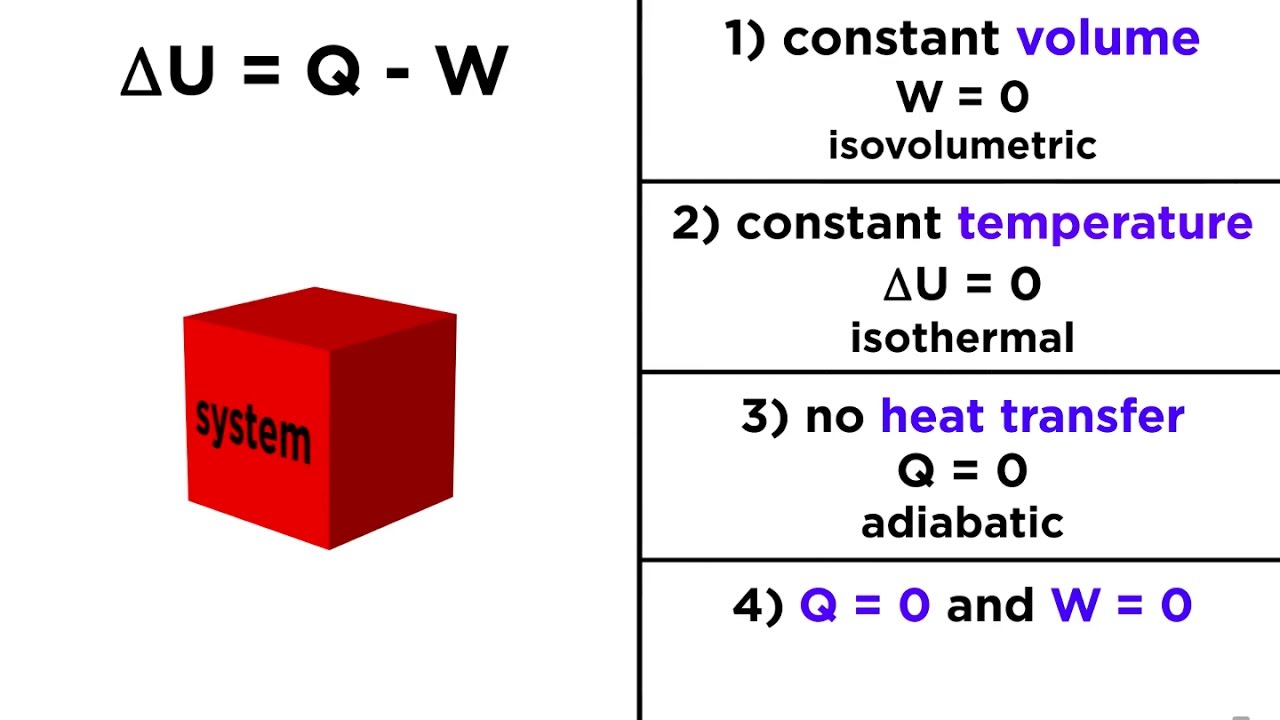
Laws of thermodynamics - Wikipedia
It can be formulated in a variety of interesting and important ways. If as a result of an exothermic reaction some energy is released to a system, it has to show up in some measurable form in terms of the state variables. Furthermore, the system either does work on it surroundings or has work done on it by its surroundings. In terms of this quantity it implies that When two initially isolated systems in separate but nearby regions of space, each in thermodynamic equilibrium with itself but not necessarily with each other, are then allowed to interact, they will eventually reach a mutual thermodynamic equilibrium. A closed system may still exchange energy with the surroundings unless the system is an isolated one, in which case neither matter nor energy can pass across the boundary.
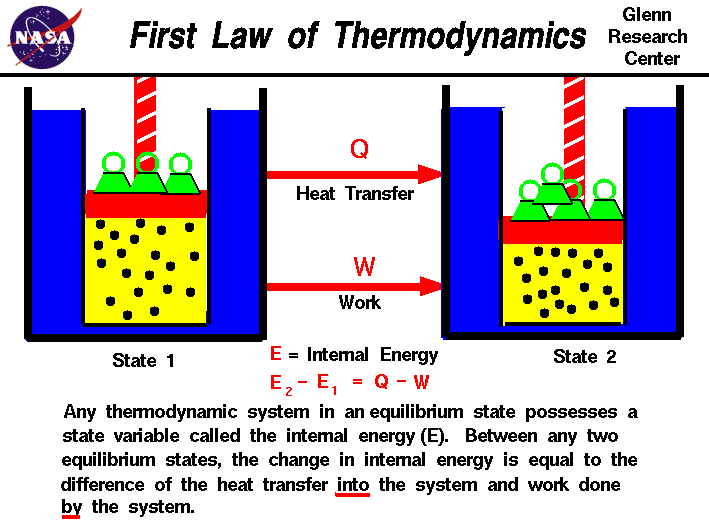




First law of thermodynamics

Description: The latter of these is what we call heat. Under these conditions, the heat given off or absorbed by the reaction would be equal to the change in the internal energy of the system. We will therefore abbreviate the relationship between the enthalpy of the system and the internal energy of the system as follows. Heat removed from a system would be assigned a negative sign in the equation. Kinetic energy is energy in use or motion.
Views: 4376
Date: 26.10.2015
Favorited: 5

































User Comments
Post a comment
Comment: